Methanol (methyl alcohol) was recommended to me many years ago, as both being harmless to brake materials, and dissolving the glycol. When I did the TR3A conversion, I did replace all the seals and flush everything with methanol. No indication that there was any glycol remaining in the system. Note please that there are lots of kinds of "alcohol" around, methanol is NOT the same as rubbing alcohol, denatured alcohol, white lightning, etc.
For all the other cars I've converted, I've just not worried about it. Yes, the glycol & silicone remain separate. But they are fully compatible with each other, neither causes the other to deteriorate any faster than it would otherwise. What happens is that any remaining glycol tends to vacuum up water, debris from worn seals, etc. and of course it continues to deteriorate as it always does, so that many years later you may find some black goop in 'dead' areas (like the bottom of brake slaves). In my experience it has not been a problem.
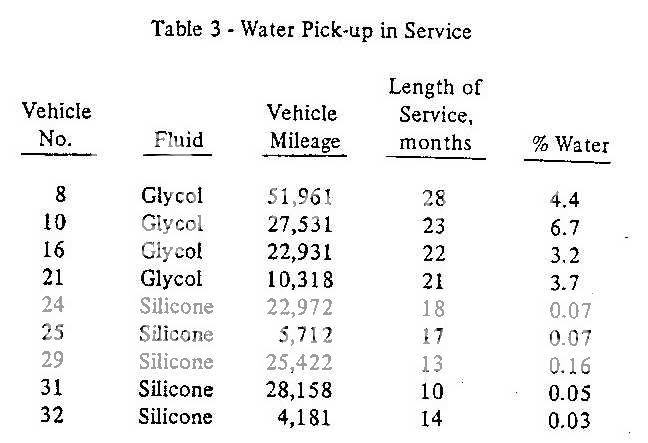
I'm suspicious of one thing in the link to the RR article above; he says that water content in glycol systems tends to maximize at 3%. That seems to be a direct contradiction to a paper presented to the SAE, which examined several "real world" examples of cars on the street, and found water contents as high as 11.1% ! This, BTW, is far above the concentration used for the "wet" boiling point specification.
It also neglects another finding from that paper, that glycol can suck not only water, but salt, right through soft brake lines. I believe this explains some of why it seems to promote corrosion so much in Triumph cylinders with aluminum bodies and steel pistons.
Interesting comment about corrosion ... My experiments show that even with deliberately introduced water, the silicone surrounds the water and keeps it out of contact with the metal. This is the "barrier film" referred to in the above article. Of course, adding water to brake systems is something I try to avoid, so it's no wonder I've never seen any corrosion from it in practice


 Hey Guest!
Hey Guest!

 smilie in place of the real @
smilie in place of the real @
 Pretty Please - add it to our Events forum(s) and add to the calendar! >>
Pretty Please - add it to our Events forum(s) and add to the calendar! >> 





 Another possibility is that the requirements that originally led to the development and adoption of DOT 5 no longer apply. Legend has it that the requirement was to be able to store vehicles at remote, unattended locations for years or even decades and have them ready to go at a moments notice. DOT 3/4 is clearly incompatible:
Another possibility is that the requirements that originally led to the development and adoption of DOT 5 no longer apply. Legend has it that the requirement was to be able to store vehicles at remote, unattended locations for years or even decades and have them ready to go at a moments notice. DOT 3/4 is clearly incompatible: